3. Kontopoulos, D.-G.✉, Patmanidis, I., Barraclough, T., and Pawar, S. (2024). Changes in flexibility but not in compactness underlie the thermal adaptation of prokaryotic adenylate kinases. bioRxiv 2024.09.04.611173. 
Abstract:
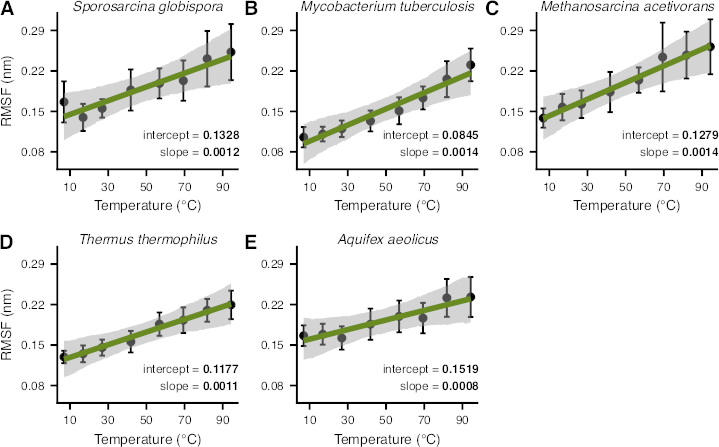
Understanding the structural changes that enable enzymes to remain active in extreme thermal conditions is of broad scientific interest for both fundamental and applied biological research. Three commonly discussed mechanisms that underlie the thermal adaptation of enzymes include modifications in structural flexibility, compactness, and in the contact network among amino acids. However, most previous studies on these topics have been limited to small sample sizes or a narrow taxonomic focus, and the relative importance of these factors to thermal adaptation remains poorly understood. In this study, we combined molecular dynamics simulations and phylogenetic comparative analyses to thoroughly analyse the structural factors underlying thermal adaptation across 70 prokaryotic adenylate kinases, a key enzyme involved in cellular energy balance and homeostasis. We detect systematic increases in the flexibility of the enzyme with temperature, both across and within species. In contrast, structural compactness appears to be almost completely independent of temperature. Finally, we detect a remarkable diversity in the contact networks of different adenylate kinases that cannot be explained solely by temperature. Our results suggest that there are multiple paths toward the adaptation of prokaryotic adenylate kinases to extreme thermal environments, but such paths generally involve changes in flexibility.